 Hydrogen bonds The helical structure of proteins or the alpha helix is the secondary structure of proteins and it is stabilized by hydrogen bonds. What bond stabilizes helical protein structure? Sheets are formed when several -strands self-assemble, and are stabilized by interstrand hydrogen bonding, leading to the formation of extended amphipathic sheets in which hydrophobic side-chains point in one direction and polar side-chains in the other (Fig. Q1: Which of the following does not affect the stability of a -helix? Explanation: The occurrence of Proline and Glycine residues affect the stability of a -helix. Which does not affect the stability of an alpha helix? The alpha-helix is a right-handed helical coil that is held together by hydrogen bonding between every fourth amino acid. Tertiary structure is the overall shape of a polypeptide, which may be stabilized by hydrophobic interactions, hydrogen bonds, ionic bonds, and peptide bonds. 
Secondary structures, which include the alpha helix or beta pleated sheet, are held together by interactions between R groups. Read More: How do you abbreviate International Immunopharmacology? Which type of interaction stabilizes the helix and the pleated sheet structures of proteins quizlet? Hydrogen bonding is responsible for the formation of alpha-helix and beta-sheet structures in proteins. Hydrogen bonds (C) The hydrogen bonds between peptide groups. Which type of interaction stabilizes the alpha helix and B pleated sheet structure of proteins? In -helix structure of proteins, the polypeptide chains are stabilized by intramolecular hydrogen bonding whereas -pealed sheet structure of proteins is stabilized by intermolecular hydrogen bonding. Which type of interaction stabilizes the alpha coiled helix and the beta folded pleated sheet structures of proteins? In particular, the CO group of each amino acid forms a hydrogen bond with the NH group of the amino acid that is situated four residues ahead in the sequence (Figure 3.30). The helix is stabilized by hydrogen bonds between the NH and CO groups of the main chain. What interactions stabilize folding of an alpha helix? A right-handed helix is most stable for L-amino acids. The -helix is very stable because all of the peptide groups (CONH) take part in two hydrogen bonds, one up and one down the helix axis. What are alpha helices and beta sheets stabilized by?īoth alpha helices and beta sheets are stabilized by hydrogen bonds. … Another factor affecting -helix stability is the total dipole moment of the entire helix due to individual dipoles of the C=O groups involved in hydrogen bonding. 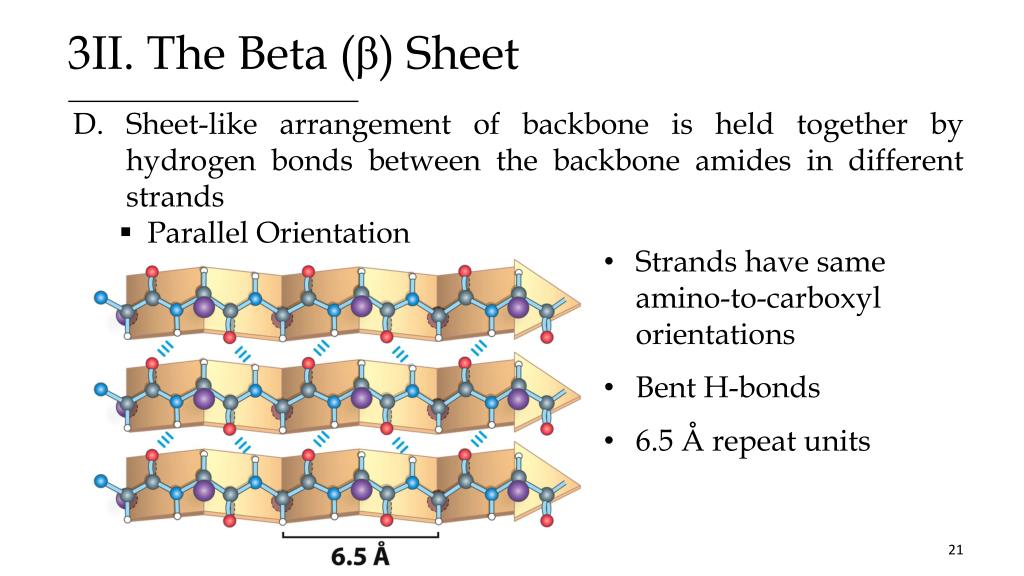
What affects alpha helix stability?Īn -helix is a right-handed coil of amino-acid residues on a polypeptide chain, typically ranging between 4 and 40 residues. Hydrogen bonds Alpha-helix is stabilized by hydrogen bonds between carbonyl residue of amino acid at position N th and amine residue of amino acid at position N+4 th. Which type of reaction stabilizes the alpha helix? It is stabilized by the regular formation of hydrogen bonds parallel to the axis of the helix they are formed between the amino and carbonyl groups of every fourth peptide bond. The -helix is a right-handed helix with the peptide bonds located on the inside and the side chains extending outward. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
